How to Track Nootropic Effectiveness
After eighteen years of systematic personal experimentation with nootropics, the single biggest mistake I see in the community isn’t choosing the wrong compounds — it’s failing to measure anything properly. People begin a new nootropic, feel subjectively better for a few days, declare it working, and move on. Or they feel nothing in week one, conclude it’s ineffective, and discontinue something that might have produced real results at week eight. In both cases, the outcome was determined not by the compound but by the absence of a methodology.
This matters enormously because the placebo effect in cognitive enhancement research is remarkably potent. Research published in Frontiers in Psychiatry found that expectation of receiving a cognitive-enhancing substance alone — without any active compound — produced measurable improvements in actual attention performance. A study from PNAS demonstrated that participants who believed they were in a cognitive training study showed significant fluid intelligence gains equivalent to 5–10 IQ points — from expectation alone. Without a rigorous personal testing protocol, you are conducting an anecdote, not an experiment.
The 4-week protocol I’m going to describe is the exact methodology I have refined and used for every nootropic trial I’ve run over the past two decades. It’s designed to be practical enough for daily life while rigorous enough to produce data you can actually trust. Before diving in, I’d recommend familiarising yourself with the broader context of how nootropics work in my Nootropics & Supplements guide — understanding mechanisms makes your observations sharper and more interpretable.
Why Most Nootropic Self-Testing Fails (And What to Do Instead)
The core problem with informal nootropic self-assessment is that human cognition fluctuates dramatically based on variables that have nothing to do with the compound being tested. Sleep quality, hydration, caffeine timing, stress levels, menstrual cycle phase, ambient temperature, time of day, and even how interesting your work is on a given day all meaningfully affect how sharp and focused you feel. Without controlling for or tracking these variables, you will inevitably attribute their effects — positive or negative — to your nootropic.
The second failure mode is the absence of a baseline. This is so fundamental that it deserves emphasis: if you don’t know where you started, you cannot know where you’ve arrived. The majority of people I’ve spoken with over the years who “know” a particular nootropic works for them have never established a pre-supplementation baseline against which to compare their current performance. What they’ve actually measured is their cognitive performance while taking a nootropic — which tells them nothing about whether the nootropic is responsible for any particular level of performance.
The third problem is over-reliance on subjective feeling. “I feel sharper” is not data. It’s a starting point for a hypothesis. Feeling sharper correlates with being sharper only loosely, and can be generated entirely by expectation, improved mood, reduced anxiety, or any number of other variables. The protocol below addresses all three of these failure modes systematically.
The Four Components of a Reliable Personal Testing Protocol
A robust self-testing protocol requires four elements working in concert: standardised objective testing, validated subjective daily ratings, confound variable tracking, and a pre-defined decision framework. Each element compensates for the weaknesses of the others.
Component 1: Standardised Objective Cognitive Testing
The most significant upgrade you can make to personal nootropic testing is replacing informal subjective assessment with standardised cognitive tests. The gold standard platform for accessible, scientifically-validated online cognitive assessment is Creyos (formerly Cambridge Brain Sciences), developed by neuroscientist Professor Adrian Owen. The platform has been used in over 400 peer-reviewed studies and maintains a normative database from over 12 million completed cognitive tests — meaning your results are benchmarked against a validated population, not just your own subjective impression.
Research from the Owen lab confirms that unsupervised, web-based Cambridge Brain Sciences assessments produce results statistically comparable to in-laboratory neuropsychological testing — which means you can generate clinically-meaningful cognitive data from home, at no cost beyond your time. The individual tasks cover short-term memory, reasoning, attention, and verbal ability — the four domains most relevant to nootropic assessment. Each task takes 1.5–3 minutes.
There is one important caveat: practice effects. Performing the same cognitive tests repeatedly will produce score improvements from familiarity rather than genuine cognitive enhancement. This is why the protocol includes a familiarisation period before baseline data collection begins — consistent with the methodology used in controlled nootropic trials that include a familiarisation session to separate practice effects from compound effects.
Component 2: The Daily Subjective Rating System
Subjective data is not worthless — it becomes worthless only when it’s the only data. A well-structured daily rating system captures the texture of cognitive experience that objective tests miss: mental clarity, word retrieval speed, creative fluency, resistance to distraction, and motivational engagement. Rated consistently and cross-referenced against objective scores, subjective data significantly improves interpretation.
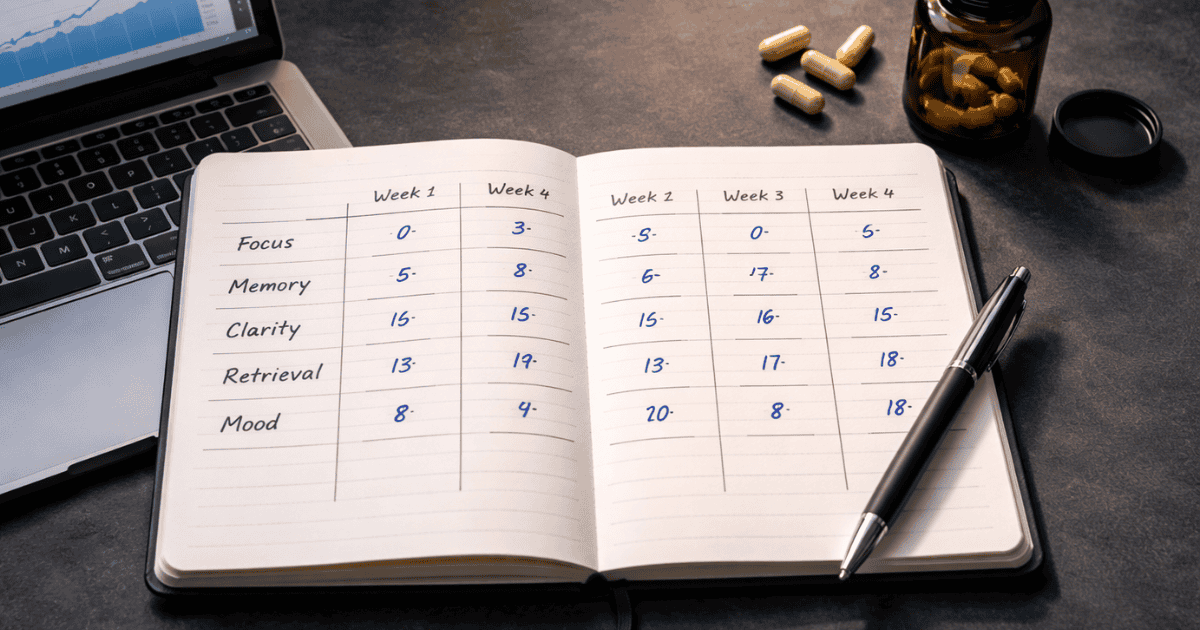
The rating system I use scores five dimensions on a 1–10 scale, completed each evening at a consistent time. Evening rating matters because it captures the full day’s cognitive experience, not just the immediate post-dose window that morning ratings tend to reflect. The five dimensions are: Focus quality (ability to sustain deep attention on demanding tasks), Working memory (ease of holding and manipulating information), Mental clarity (absence of brain fog, sharpness of thinking), Word retrieval (speed and ease of accessing vocabulary and concepts), and Emotional baseline (anxiety, stress, mood stability — because these confound cognitive ratings significantly if not tracked separately).
Component 3: Confound Variable Tracking
This is the most frequently omitted element of personal nootropic testing, and its absence invalidates much of the data people collect. The confound variables that most reliably corrupt nootropic self-assessment data are sleep duration and quality, total caffeine intake and timing, physical exercise (aerobic exercise acutely elevates BDNF and can produce cognitive effects indistinguishable from some nootropics), alcohol consumption, major stressors, and illness. You don’t need to control all of these perfectly — that’s not feasible in real life — but you do need to record them so that anomalous data points can be identified and contextualised rather than attributed to your compound.
The daily confound log takes under two minutes. Sleep hours and a 1–5 quality rating, yes/no for exercise, total caffeine estimate, and a brief note flag for anything unusual (stressful meeting, poor night, social drinking). This doesn’t need to be a detailed diary — just enough data to explain outliers when you review your 28-day dataset. The relationship between sleep and cognitive performance is worth emphasising here: the sleep and cognitive performance connection is so robust that poor sleep will consistently outweigh any nootropic benefit — meaning you cannot meaningfully test nootropic effectiveness during a period of disrupted sleep.
Component 4: A Pre-Defined Decision Framework
One of the most insidious biases in self-testing is motivated interpretation — unconsciously reading positive signals into ambiguous data because you want the compound to work, or prematurely concluding failure because you expected faster results. The solution is to define your decision criteria before the trial begins, not after you’ve seen the data. What constitutes a positive result? A negative one? What will you do at the 28-day review? Having written answers to these questions before starting is what separates disciplined self-experimentation from expensive guesswork.
The 4-Week Protocol: Week by Week
Familiarisation & Baseline Establishment
No nootropic taken this week. This is non-negotiable and the most commonly skipped step. Week 1 serves two purposes: familiarising yourself with the cognitive tests to eliminate practice effects from your baseline data, and establishing what your “unenhanced” cognitive performance actually looks like under your normal lifestyle conditions.
Daily actions: Complete your chosen Creyos cognitive task battery each morning at the same time (within a 30-minute window). Complete your 5-dimension evening rating. Log confound variables. By day 4–5, practice effects will have plateaued and your scores will begin to reflect your genuine baseline. Days 5–7 scores become your baseline dataset. Define your decision criteria in writing: what constitutes a positive result at 28 days?
Introduction Phase — Single Compound Only
Introduce your nootropic on Day 8 at the lowest recommended dose. This week is specifically not about noticing effects — it’s about establishing a stable dosing pattern and identifying any immediate adverse reactions before committing to the full trial.
Critical rule: Change nothing else during the trial period. Same caffeine routine, same sleep habits, same exercise schedule. Every change you make introduces a confounding variable. Continue daily cognitive testing and evening ratings. Do not review or analyse your data mid-week — you’re collecting, not interpreting. Expect nothing from slow-build compounds like Lion’s Mane or Bacopa this week.
Continued Dosing — Mid-Trial Review
Continue the same dose and protocol. At the end of Week 3, conduct your first interim data review. Compare Week 3 averages against your Week 1 baseline across both objective test scores and subjective ratings. You’re looking for direction of change, not magnitude — at this stage you’re identifying whether any signal is emerging.
Interpretation guide: Consistent improvement in 3 or more dimensions with stable confound variables = promising signal. Improvement in subjective ratings but flat objective scores = possible placebo effect or mood/anxiety benefit (still potentially valuable). Flat or declining scores with no confound explanation = no effect at this dose, or compound not suited to you. Mixed or inconsistent data = continue to Week 4 without changing anything.
Final Data Collection & Decision Day
Continue dosing. At the end of Day 28, conduct your full data review. Calculate weekly averages for all five subjective dimensions and compare Week 4 scores against your Week 1 baseline. Review your objective cognitive test trend line. Cross-reference any improvements or declines against your confound log — are there lifestyle explanations for any patterns you’re seeing?
Decision framework: Apply the criteria you defined before the trial. If the result is positive, continue for a further 8 weeks before final evaluation (particularly relevant for Bacopa and Lion’s Mane, whose full effects require 12 weeks). If neutral or negative with good confound control, the compound is likely not producing meaningful effects at this dose — consider a dose increase, a different extract form, or discontinuation.
The Confound Variables That Corrupt Most Self-Tests
In nearly two decades of personal experimentation, I’ve found three confound variables responsible for the majority of misattributed nootropic effects. Understanding them transforms how you interpret your own data.
Sleep is the dominant variable. A single night of sleep debt produces cognitive impairments that exceed the effects of legal blood alcohol levels — while a night of unusually good sleep can make almost any supplement appear to be working spectacularly. This is why compounds should never be trialled during periods of disrupted sleep. If your confound log shows three or more nights below your normal sleep duration in a given week, that week’s cognitive data is unreliable for nootropic assessment purposes. The relationship between sleep quality and memory consolidation is covered in depth in my Memory & Learning Enhancement hub.
Stress changes everything. Chronic stress acutely impairs working memory, reduces cognitive flexibility, and degrades attention — the three domains most nootropic trials aim to assess. A difficult work period coinciding with the start of a new nootropic trial will reliably suppress your scores and produce a false negative result. Conversely, a period of unusually low stress will elevate scores independently of any supplementation. Your emotional baseline rating exists specifically to flag these periods so they can be weighted appropriately in your analysis.
Caffeine timing variation produces dramatic acute effects. Consuming caffeine 30 minutes earlier or later than usual, or changing your dose slightly, will register in your cognitive testing scores in ways that can entirely mask or mimic a nootropic effect. During a testing period, keep your caffeine intake — dose, timing, and source — as consistent as possible, and record any deviations. This applies even to compounds like L-theanine, which you should only trial in conjunction with consistent caffeine or entirely without it, not with varying caffeine inputs.
How to Interpret Your 28-Day Data Honestly
After 28 days of consistent data collection, you’ll have approximately 140 individual data points — enough to identify meaningful patterns while remaining humble about the limitations of an n=1 dataset. Here is how I approach the final analysis.
First, calculate weekly averages for each dimension, not daily scores. Single days are too noisy — weekly averages smooth out day-to-day variation and reveal genuine trends. Compare Week 4 averages to your Week 1 baseline and express them as percentage changes. A consistent 10% improvement in focus quality with stable confounds is a meaningful signal. A 2–3% improvement with variable confounds is noise.
Second, look for concordance between objective and subjective data. If your Creyos scores improve and your subjective ratings improve and your confounds are stable, that’s a strong positive signal. If only your subjective ratings improve, you may be experiencing a mood or anxiety benefit (which is still valuable — don’t dismiss it) or a placebo effect. If only your objective scores improve while subjective ratings are flat, the compound may be working at a neurochemical level without producing a subjectively noticeable effect — also worth continuing.
Third, and this is important: a 28-day trial is not sufficient for slow-build compounds. For Lion’s Mane, Bacopa, and other neuroplasticity or synaptic adaptation compounds, treat 28 days as a preliminary signal, not a verdict. The evidence for both Lion’s Mane and Bacopa consistently shows the most significant effects at 8–12 weeks. A positive signal at 28 days is reason to continue; a flat signal at 28 days with a slow-build compound is not reason to stop.
Key Takeaways
Frequently Asked Questions
Includes a pre-built 4-week tracking template, daily rating scorecard, and the exact cognitive testing schedule I’ve used for 18+ years of personal experimentation.
Join 2,000+ readers optimising their cognitive performance. Unsubscribe anytime.
- Dockree, S., et al. (2019). Placebo- and nocebo-effects in cognitive neuroenhancement: when expectation shapes perception. Frontiers in Psychiatry, 10, 437. PMC6640161
- Foroughi, C.K., et al. (2016). Placebo effects in cognitive training. Proceedings of the National Academy of Sciences, 113(27), 7470–7474. PNAS 10.1073/pnas.1601243113
- Timmins, M.A., et al. (2020). Comparing web-based and lab-based cognitive assessment using the Cambridge Neuropsychological Test Automated Battery: a within-subjects counterbalanced study. JMIR Mental Health, 7(8), e15792. PubMed 32749999
- Taler, V., & Phillips, N.A. (2019). Thirty-five years of computerized cognitive assessment of aging — where are we now? Journal of Alzheimer’s Disease, 70(s1), S1–S12. PMC6787729
- Medrano, M., et al. (2022). Acute effect of a dietary multi-ingredient nootropic as a cognitive enhancer in young healthy adults: a randomized, triple-blinded, placebo-controlled, crossover trial. Frontiers in Nutrition, 9, 858910. DOI: 10.3389/fnut.2022.858910
- Suliman, N.A., et al. (2022). Nootropics as cognitive enhancers: types, dosage and side effects of smart drugs. Nutrients, 14(16), 3367. PMC9415189